We are the trusted legal marketing team.
Proven Success in Legal Blogging
We Put Legal Infographics on the Map


legal website design services
Law Firm Web Design
Lawyers & law firms need a website that is updated, informative, and professional. Your website should separate you from your competitors and generate relevant leads. We build custom legal websites that are unique, powerful, and fully responsive to meet the demands and expectations of clients today.

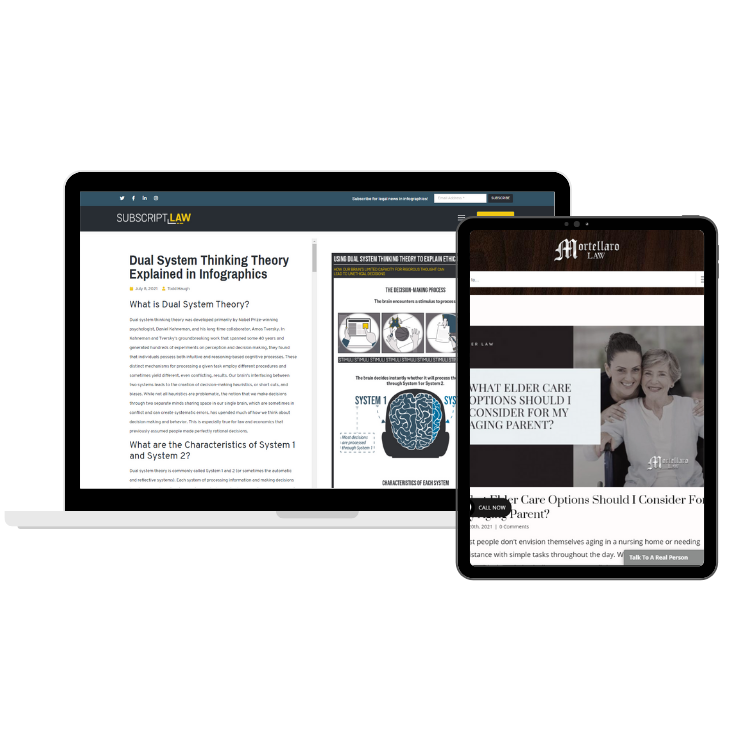
Legal Website Content Services
Web Content & SEO
Our team of legal experts is uniquely equipped to service lawyers and law firms across the country. Our team of creative designers will work to understand your firm, your requirements, and your preferred styles to create original content. Communication and support are at the core of our services to ensure you have the best experience possible.
Looking for legal marketing services for your law firm or individual practice?
We create professional & versatile infographics.
Infographics enhance your blog, email, and other marketing content. Create more engaging content with custom visual designs made uniquely for your firm.
The Latest Legal News & Updates from the Subscript Law

Dobbs v. Jackson Women’s Health
How will the Court side on this copyright case against the clothing store H&M?

Unicolors v. H&M
How will the Court side on this copyright case against the clothing store H&M?
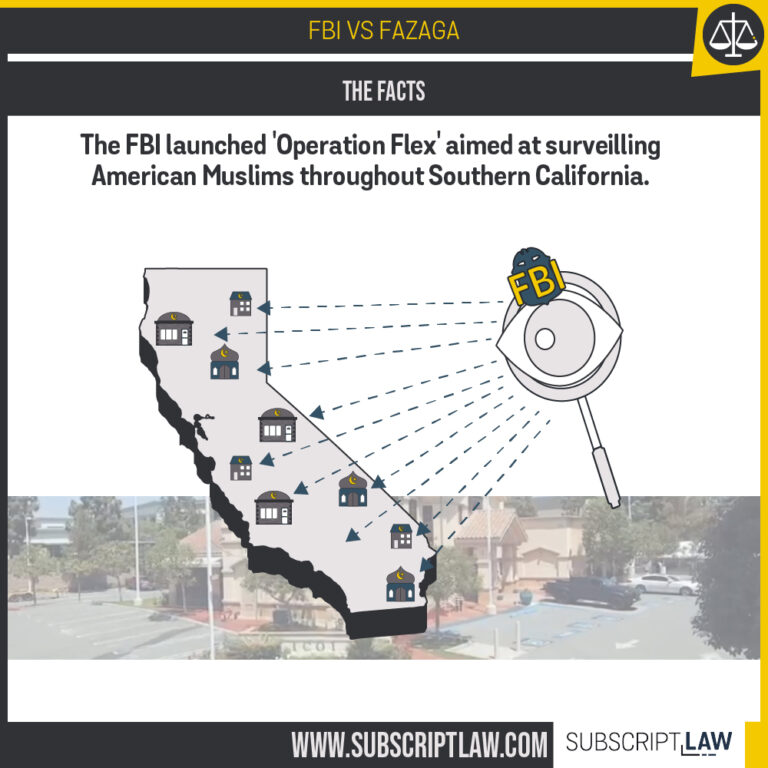
FBI v. Fazaga
Does the state secrets privilege apply to keep CIA contractors from testifying in Polish legal proceedings?
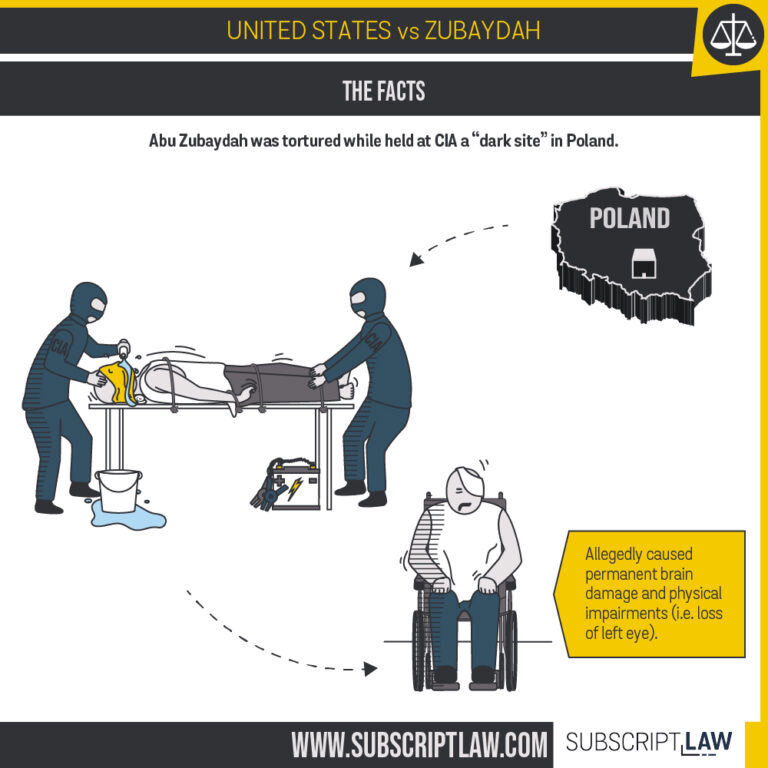
United States v. Zubaydah
Does the state secrets privilege apply to keep CIA contractors from testifying in Polish legal proceedings?
Essential SCOTUS Live Feed
The Supreme Court will let stand the conviction of a Louisiana inmate who was convicted of murder and sentenced to death for his role in an attempted prison escape involving five other inmates.
https://cnn.it/40Y3hTA
Democrats weigh trying to force Supreme Court to adopt ethics rules https://www.washingtonpost.com/politics/2023/04/03/democrats-weigh-trying-force-supreme-court-adopt-ethics-rules/
In Mexico, a reproductive rights network is helping U.S. women obtain abortions after the Supreme Court moved to eliminate the constitutional right to abortion last year. https://nbcnews.to/3Mdk154
Overruling Roe was only part of the Supreme Court's GOP agenda. Get ready for some retrograde decisions. (via Deadline: Legal Blog) https://www.msnbc.com/deadline-white-house/deadline-legal-blog/supreme-court-cases-2023-rcna64600?cid=sm_npd_ms_tw_ma&taid=642ad4def30e380001d83563&utm_campaign=trueanthem&utm_medium=social&utm_source=twitter
His obsessive online posts terrorized her. But were they illegal threats? The tale of the people at the center of a Supreme Court case will test limits of First Amendment protections https://www.washingtonpost.com/politics/2023/04/01/scotus-true-threats-coles-whalen/
“The scarcity of experienced civil-rights attorneys is a direct result of how these attorneys are paid—and, in many cases, not paid—for their work,” @JCSchwartzProf writes. http://on.theatln.tc/PFoAxaH
“The scarcity of experienced civil-rights attorneys is a direct result of how these attorneys are paid—and, in many cases, not paid—for their work,” @JCSchwartzProf writes. http://on.theatln.tc/qArABTE
Voters will decide Tuesday if conservative or liberal judges control the Wisconsin Supreme Court ahead of decisions on abortion and redistricting. https://wapo.st/3zojNjE
Wisconsin voters head to the polls Tuesday to elect their next state Supreme Court justice in what could be the most consequential race of 2023. https://cbsn.ws/3M6r1AD
We’ve seen the Supreme Court trample rights under the guise of originalism. That's why Thomas' critique of this historic ruling is worrisome. (via Deadline: Legal Blog) https://www.msnbc.com/deadline-white-house/deadline-legal-blog/clarence-thomas-gideon-wainwright-precedent-rcna75535?cid=sm_npd_ms_tw_ma&taid=64287f581f4ecb0001b65e42&utm_campaign=trueanthem&utm_medium=social&utm_source=twitter
The Supreme Court rejected a wild appeal that put separation of powers involving prosecutors in the spotlight. Justices Gorsuch and Kavanaugh objected. (via Deadline: Legal Blog) https://www.msnbc.com/deadline-white-house/deadline-legal-blog/gorsuch-kavanaugh-prosecutorial-discretion-trump-rcna76803?cid=sm_npd_ms_tw_ma&taid=64286a26de7fbf000151c5da&utm_campaign=trueanthem&utm_medium=social&utm_source=twitter
Join #NCCed for a fast-paced and fun top-15 SCOTUS case review, a typical course of study for an #APGov class.
View the schedule and register: http://ow.ly/Ek0250NqqGG
Johnson & Johnson unit loses bid to stay in bankruptcy during Supreme Court appeal http://reut.rs/3M3MIkM
Sarah Posner: Ginni Thomas has become the face of the Supreme Court's ethics issues. But that ignores a much bigger problem. https://www.msnbc.com/opinion/msnbc-opinion/supreme-court-ginni-thomas-clarence-thomas-donations-rcna77286?cid=sm_npd_ms_tw_ma&taid=64278ca65e1c3000019a21f4&utm_campaign=trueanthem&utm_medium=social&utm_source=twitter
Trump indictment unlikely to sway Wisconsin Supreme Court race https://fxn.ws/3znKo0k
Interested in becoming a contributor?
We’re on the lookout for lawyers who share our passion for teaching legal issues. Write a report on a Supreme Court case or legal topic of your expertise. We’ll provide the infographic, and you’ll get the recognition.

